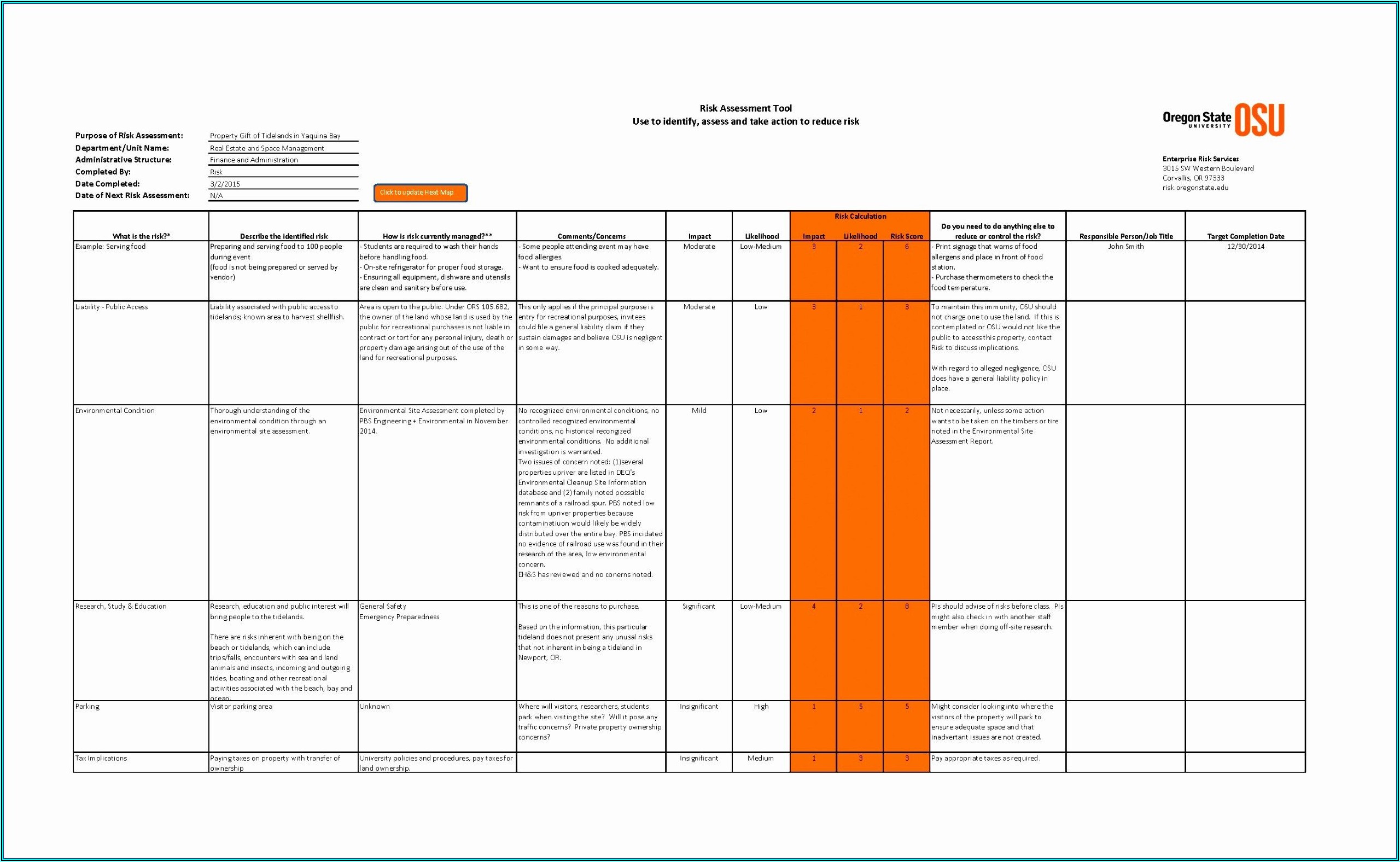
arrangements for review and approval of the plan arrangements for conducting the evaluation arrangements for gathering applicable information from the published literature In accordance with ISO 10993-1 (2018), as well as the trends of the European authorities ( EU MDR 2017/745) and the FDA, physical and/or chemical characterization shall precede any biological test in the biological evaluation plan. Thus, the biological evaluation plan is a crucial first step in the biocompatibility assessment of a medical device the plan details the overall strategy to prove the biocompatibility of medical devices within a risk management process. The biological evaluation shall be planned, carried out, and documented by knowledgeable and experienced professionals.”. This risk management process involves identification of biological hazards, estimation of the associated biological risks, and determination of their acceptability.  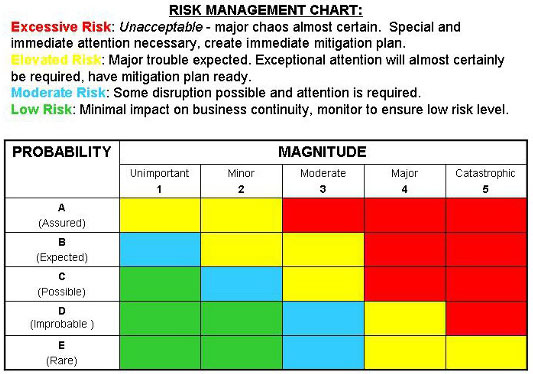
According to ISO 10993-1 (2018): “The biological evaluation of any material or medical device intended for use in humans shall form part of a structured biological evaluation plan within a risk management process in accordance with ISO 14971:2007 (the standard has been revised by ISO 14971:2019).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
